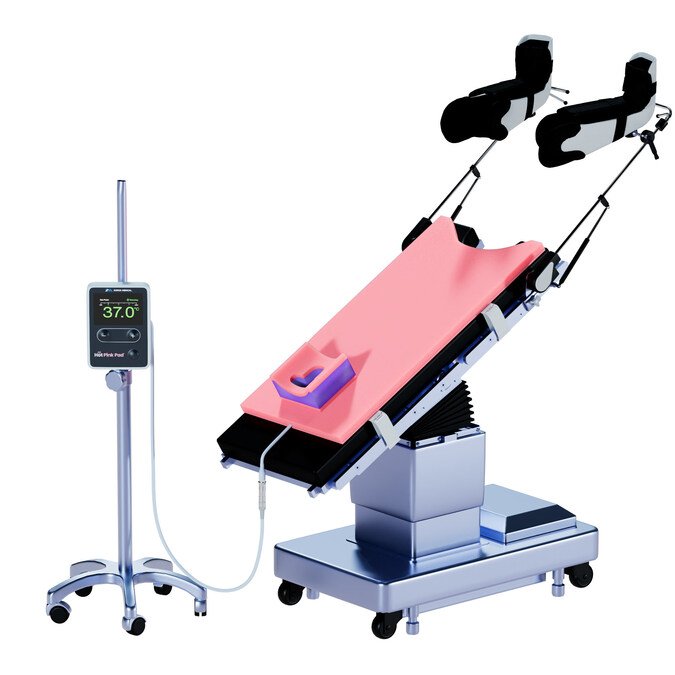
A next-generation surgical positioning system has received FDA 510(k) clearance in the United States, introducing an integrated approach to patient positioning and temperature management during surgery.
Developed by Xodus Medical, the Hot Pink Pad builds on existing operating room positioning technologies by combining non-slip support with thermal regulation capabilities. The system is designed to address two critical intraoperative challenges: maintaining stable patient positioning and preventing hypothermia.
Surgical positioning systems such as the Pink Pad are used to stabilise patients during procedures requiring steep positioning, including Trendelenburg orientation, where patients are tilted to improve surgical access. These systems help prevent patient movement, distribute body weight, and reduce the risk of pressure-related injuries during prolonged procedures.
The newly cleared system expands on this concept by incorporating temperature management, which plays a key role in surgical outcomes. Maintaining normothermia during surgery is critical to reducing complications such as infection risk, delayed recovery, and extended hospital stays.
The Hot Pink Pad also reflects a broader trend toward integrating multiple safety functions into a single device, reducing the need for separate equipment in the operating room. By combining positioning and thermal support, the system aims to streamline workflows while improving overall patient care.
Designed for compatibility with existing surgical setups, the system supports ease of use in high-intensity operating environments. Its materials are engineered to provide both stability and pressure redistribution, helping minimise risks such as patient sliding and hospital-acquired pressure injuries.
The FDA 510(k) clearance confirms that the device meets safety and performance standards for clinical use in the United States, supporting its commercial launch and adoption across surgical centres.